Editor’s Note: Unfortunately disruption has made this edition of The Badger a little different. Unavoidable circumstances meant that this edition was delayed. Consequently, some of these articles are a little older and we haven’t been able to get the newest stories to you this time. However, we think that it is fair that those who wrote great articles have them published, and these important stories are read by you.
Words by Melis Trimmer, Staff Writer
Trigger warnings for weight loss and drug use.
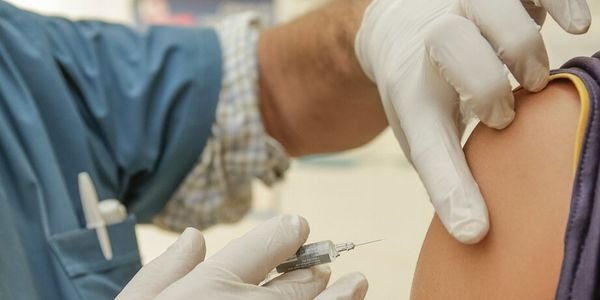
On February 8th 2022, The National Institute for Health and Care Excellence (NICE) issued a draft guidance recommending Semaglutide, also known as Wagovy – an anti-diabetic drug, as a medication to tackle weight loss. The drug will be available on the NHS for adults with either; at least one weight-related condition such as obstructive sleep apnoea, or heart disease, and a BMI of at least 35kg/m2, or, alternatively those with a BMI of 30.0 kg/m2 to 34.9 kg/m2 if they are referred to tier 3 services based on the criteria in NICE’s clinical guideline on obesity: identification, assessment and management.
Semaglutide is a drug administered once weekly, in the form of an injection and works by mimicking the hormone glucagon-like peptide 1 (GLP-1) which we release naturally after eating. The drug increases insulin secretion, suppresses glucagon secretion and slows gastric emptying. Patients will inject themselves with Semaglutide, which will work to trick the brain into thinking they have eaten, suppressing appetite and therefore losing weight. NICE have reported that patients already on the weekly injections have seen their weight fall by an average of 12% after one year.
NICE’s independent appraisal committee recommended that Semaglutide can only be prescribed as part of a specialist weight management service for a maximum of two years, alongside a reduced-calorie diet and increased physical activity.
Government estimates indicate that the current costs of obesity in the UK are £6.1 billion to the NHS. The 2019 Health Survey for England estimated that 28% of adults in England were obese and a further 36% were overweight. Given these figures, the drug has been stated by experts to be a “gamechanger” for tackling the obesity crisis in England, which has been addressed previously by the government. In 2020, Prime Minister Boris Johnson launched the anti-obesity strategy which included a ban of TV and online adverts for foods high in fat, sugar and salt before 9pm and ending deals such as buy-one-get-one-free on unhealthy foods high in salt, sugar and fat.
Helen Knight, programme director in the centre for health technology evaluation at NICE, said: “We know that management of overweight and obesity is one of the biggest challenges our health service is facing with nearly two thirds of adults either overweight or obese. It is a lifelong condition that needs medical intervention, has psychological and physical effects, and can affect quality of life,” and added “but in recent years NICE has been able to recommend a new line of pharmaceutical treatments which have shown that those people using them, alongside changes to their diet and exercise, have been able to reduce their weight.”
The list price of Semaglutide 2.4 mg and 1.7 mg is commercial in confidence and cannot be reported here. The list price of Semaglutide 0.25 mg, 0.5 mg and 1.0 mg is £73.25 per pack (4 pre-filled pens; excluding VAT).
The drug has been approved by the NHS but plans cannot be made to roll out Semaglutide until NICE’s final guidance is released.